Breast surgery is common for reconstruction after cancer or cosmetic changes like augmentation, reduction, or lifts. Surgical mesh, often acellular dermal matrix (ADM) or synthetic materials, provides tissue support but carries risks that vary by patient and technique.
What Is Breast Mesh?
Breast mesh is a material used in breast surgery to provide added support to surgical implants. Many plastic surgeons use it in select cases to reinforce tissue and improve implant positioning.
Doctors sometimes refer to mesh as an “internal bra” device because it helps support the lower portion of the breast and stabilize the implant pocket.
Mesh in breast surgery has become more common in both reconstructive surgery and cosmetic procedures such as breast augmentation and breast lift procedures. In some cases, patients may not realize that what was described as extra support involved placing breast mesh implants inside the body.
When Is Mesh Used?
In certain internal bra procedures, surgeons may use mesh to reinforce thin tissue, reduce the risk of implant displacement, or help define the implant pocket during reconstructive surgery.
Mesh is sometimes selected in higher-risk cosmetic cases such as breast augmentation with large implants or breast lift procedures where tissue support is limited.
Different mesh brands are available on the market, and outcomes may vary depending on the specific product used and the patient’s health profile.
Possible Complications
Some patients experience internal bra complications that go beyond normal surgical healing.
These problems may include implant displacement, shifting of the implant pocket, or painful complications that interfere with daily life. In more serious cases, patients report severe breast pain that does not improve with time and may signal mesh failure.
While many women heal without issue, others may develop a breast deformity caused by implant displacement or improper mesh integration.
How Common Are Breast Mesh Complications?
Studies show that mesh complications can occur in approximately 5% to 30% of cases. However, risk levels often depend on factors such as:
- Whether the surgery was done in one stage or two stages
- Smoking history
- Obesity
- Overall health
This means complications are not always caused by mesh alone. Patient health and surgical technique also play a role.
Most Reported Complications
Common complications linked to mesh in breast surgery include:
- Infection (5–10%)
- Seroma, or fluid buildup (3–15%)
- Skin necrosis (loss of skin tissue)
- Device malposition (about 9%)
- Capsular contracture (up to 16% in some groups)
These issues may require antibiotics, fluid drainage, or even revision surgery.
Less Common But Serious Problems
Less frequent complications can include:
- Nerve pain or numbness
- Mesh migration
- Implant rupture
- Imaging interference during mammograms
Patients should always inform radiologists if mesh or breast mesh implants were used.
Some research shows no overall increase in major complications when acellular dermal matrix (ADM) is used in single-stage reconstruction procedures. However, outcomes vary depending on the individual patient and the specific mesh brands used.
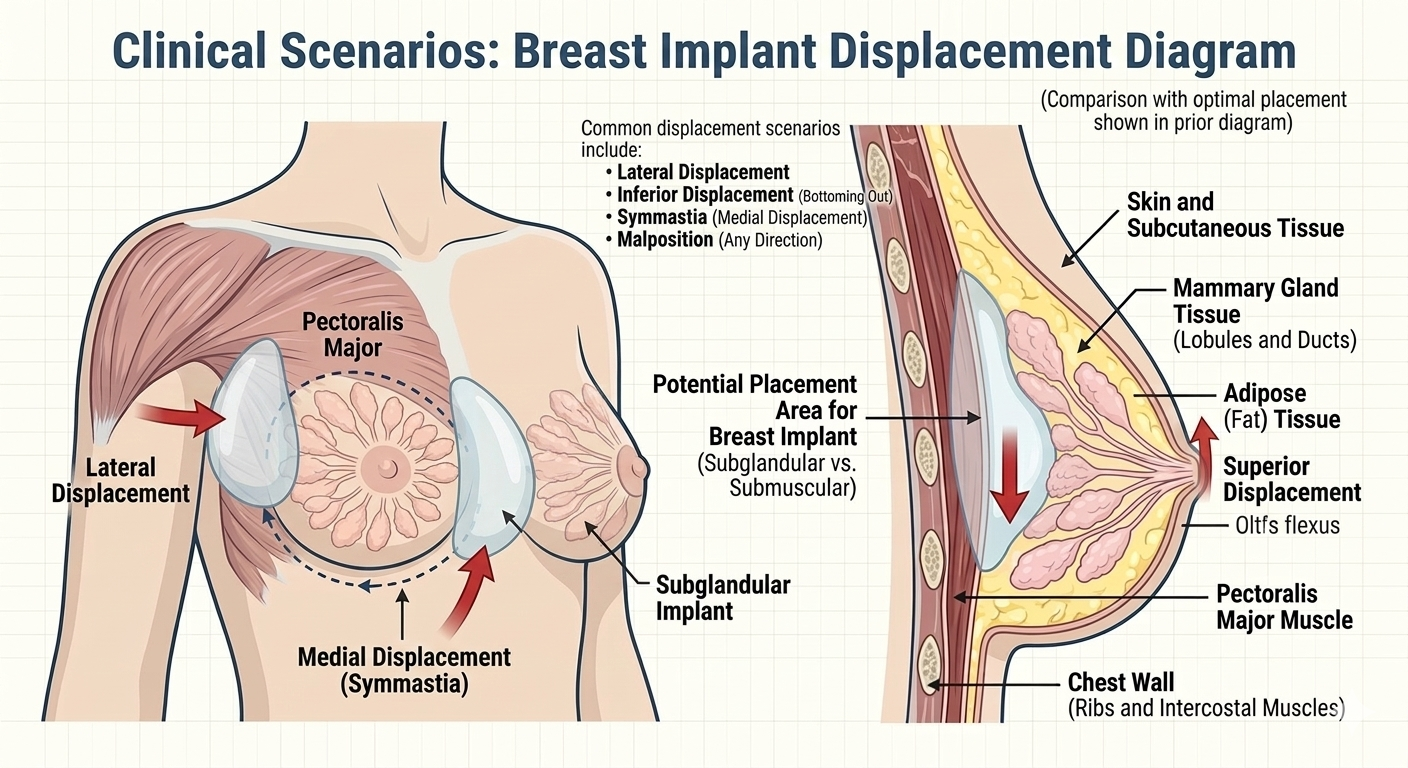
Signs of Breast Mesh Failure
Not every surgery problem means mesh failure. But some symptoms may point to a serious breast mesh complication.
Watch for:
- Ongoing swelling that doesn’t go away
- Fluid buildup (seroma) that keeps coming back
- Redness, warmth, or fever (possible infection)
- Severe or burning nerve pain
- Implant shifting or dropping
- Skin thinning or opening
- Needing implant or mesh removal
If your body is not healing correctly, the mesh may not be integrating as expected. In some cases, the mesh can harden, move, or trigger infection.
When surgical mesh does not work properly, doctors may recommend revision surgery or removal.
Internal Bra Surgery: What Patients Weren’t Always Told
The term “internal bra” sounds simple and safe. But many patients say they did not fully understand:
- A foreign material was being placed inside their body
- The long-term risks of mesh
- That removal can be complex
- That additional surgeries may be needed
An internal bra is not just stitches. It often involves ADM or synthetic mesh products placed under the breast tissue.
While many patients heal well, others experience serious complications that require more procedures.
If you were told you needed an “internal bra” and later developed infection, migration, or reconstructive failure, your case may deserve review.
Mesh in Breast Surgery: Questions Patients Should Ask
- What type of mesh or internal bra device was used?
- Which mesh brands were selected?
- Was the implant pocket reinforced with mesh?
- What are the risks of implant displacement?
- What are the signs of painful complications?
- Could mesh failure lead to reconstructive surgery again?
Asking these questions can help patients better understand their risks before and after surgery.
Understanding the Role of Plastic Surgeons and Surgical Implants
Breast mesh is typically used alongside surgical implants during cosmetic or reconstructive procedures. Plastic surgeons may recommend mesh to reinforce tissue, strengthen the implant pocket, or reduce strain on the lower portion of the breast.
However, even when performed by qualified plastic surgeons, outcomes can vary. Surgical implants rely on proper tissue healing, and when mesh does not integrate as expected, complications such as implant displacement, severe breast pain, or implant loss may occur.
It is important for patients to understand that mesh is not a guarantee against future problems. In some cases, additional procedures may be required to correct implant positioning or address painful complications.
What Is Revision Surgery?
Revision fixes issues like infection, malposition, or poor healing, often needed in 10-20% of reconstructions regardless of mesh.
Stress, recovery time, and costs rise with revisions; check records to confirm mesh use, as not all patients recall details.
Common Myths About Cosmetic Breast Mesh Procedures
Debunking Misconceptions
There is a lot of confusion online about mesh in breast surgery and internal bra procedures. Some information is helpful, but some of it is misleading. Let’s clear up a few common myths.
Myth #1: Breast Mesh Is Always Dangerous
Not true.
Many women have successful surgeries using surgical mesh or internal bra devices without serious complications. Mesh is designed to support tissue and strengthen the implant pocket. Problems can happen, but they do not happen in every case.
Like any medical device, outcomes depend on patient health, surgical technique, and the specific mesh brands used.
Myth #2: Internal Bra Procedures Are Just Stitches
Some patients believe an “internal bra” simply means extra sutures.
In reality, many internal bra procedures involve placing a mesh material inside the body. These internal bra devices are meant to act like a sling to support implants or tissue.
It is important for patients to understand whether mesh was used during their surgery.
Myth #3: All Complications Mean Mesh Failure
Not always.
Complications like swelling or discomfort can happen with any reconstructive surgery or cosmetic procedure such as breast augmentation or breast lift procedures.
However, persistent implant displacement, severe breast pain, or repeated infections may raise concerns about mesh failure or internal bra complications.
Every case must be evaluated individually.
Myth #4: Mesh Eliminates the Need for Future Surgery
Some patients are told that mesh provides long-term support and prevents future issues.
While mesh can improve stability, it does not guarantee that revision surgery will never be needed. Factors like aging, weight changes, pregnancy, or healing issues can still affect results.
Even without mesh, revision rates for reconstructive surgery can range from 10–20%.
Myth #5: Only Cosmetic Patients Experience Problems
Breast mesh implants are used in both cosmetic and cancer-related reconstructive surgery.
Complications can occur in either setting. The reason for surgery does not automatically determine the risk level.
Myth #6: If It’s FDA Approved, There Are No Risks\
FDA approval means a product met regulatory standards for use. It does not mean the product is risk-free.
All surgical mesh devices carry potential risks, including infection, migration, painful complications, or implant displacement.
That is why informed consent and clear communication are so important.
The Bottom Line
Cosmetic breast mesh procedures are not automatically good or bad. They are medical tools that can help in certain situations.
The key is understanding:
- What type of mesh was used
- Why it was selected
- What risks were discussed
- What symptoms should not be ignored
If something does not feel right after surgery, do not ignore it. Ask questions. Review your records. And seek medical advice if needed.
Being informed is the best way to protect your health and your future.
Legal Considerations
Some breast mesh lawsuits fall under product liability law, which examines whether a product was defectively designed, manufactured improperly, or marketed without proper warnings.
When mesh in breast surgery leads to severe breast pain, reconstructive surgery failure, or implant displacement, legal review may focus on whether the mesh brands used were associated with higher complication rates.
Not all internal bra procedures result in injury, but when internal bra complications lead to multiple surgeries or breast deformity, further evaluation may be appropriate.
Other Breast Mesh Lawsuits and Legal Trends
Breast mesh lawsuits are not entirely new. Over the years, legal claims involving surgical mesh devices have raised questions about product safety, manufacturer warnings, and long-term complications.
While many mesh products are used successfully, some lawsuits have alleged:
- Failure to properly warn patients about risks
- Defective design of certain mesh brands
- Increased risk of infection or implant loss
- Painful complications requiring revision surgery
- Reconstructive surgery failure
These cases often fall under product liability law, which examines whether a medical device was reasonably safe and whether adequate warnings were provided.
It is important to understand that not all mesh complications result in lawsuits. Courts typically focus on severe complications, documented medical harm, and cases involving implant loss, multiple revision procedures, or significant long-term damage.
As research and litigation continue, patients are becoming more aware of potential internal bra complications and the risks associated with mesh in breast surgery.
| Complication | Frequency with ADM | Common Management |
| Infection | 5-10% | Antibiotics, drainage |
| Seroma | 3-15% | Aspiration, monitoring |
| Capsular Contracture | Up to 16% | Revision, medication |
| Malposition | ~9% | Surgical repositioning |
Breast Mesh Lawsuit: When Does It Apply?
A breast mesh lawsuit may apply when complications go beyond normal surgical risks.
Legal cases often involve:
- Severe infections requiring hospitalization
- Abscess or tissue damage
- Mesh removal (explant)
- Reconstructive failure
- Implant rupture connected to mesh
- Multiple revision surgeries
- Long-term nerve damage
Courts look at whether the product failed, whether warnings were adequate, and whether the patient suffered measurable harm.
Not every cosmetic issue qualifies. But serious medical complications may.
If you had an internal bra lawsuit concern because you needed revision surgery after mesh placement, it is worth asking questions.
Who Qualifies for a Breast Mesh Lawsuit?
Not every complication after breast surgery qualifies for a lawsuit. However, certain situations may meet legal criteria.
Below is a general overview of who may qualify for a breast mesh lawsuit.
1. Mesh Was Used in Your Breast Surgery
You may qualify if a mesh product was implanted during an internal bra procedure in one of the following surgeries:
- Breast reconstruction after mastectomy
- Cosmetic breast augmentation
- Cosmetic breast reduction
- Cosmetic breast lift
If mesh was placed to support the implant or reinforce tissue, your case may be eligible for review.
2. You Had or Need Revision Surgery
A strong qualifying factor is whether you
- Already had revision surgery
- Have revision surgery scheduled
- Are currently discussing revision surgery with your doctor
This applies even if you are unsure whether mesh was used.
If your doctor is recommending another surgery because of complications, whether caused by mesh or possibly related to the implant, your situation may still qualify for evaluation.
3. You Experienced Serious Complications
Not all surgical issues qualify. Cases typically involve more serious medical complications, such as:
- Nerve damage
- Infection or abscess
- Seroma (fluid buildup)
- Migration or implant displacement
- Explant (implant or mesh removal)
- Reconstructive failure
- Implant rupture
- Interference with breast imaging
These complications often require medical treatment, hospitalization, or additional surgery.
Who May Not Qualify
Some cases may not meet legal criteria.
Cases are generally declined if the only reported injuries are:
- Capsular contracture
- Cosmetic disfigurement alone
- Asymmetry alone
While these issues can be upsetting, they may not qualify unless combined with more serious medical complications.
Surgery Date and Location Matter
Eligibility may also depend on timing and location.
Cases are typically declined if:
- The surgery where mesh was used (or may have been used) occurred in 2014 or earlier
- The implant surgery took place in Texas or Michigan
These factors can affect legal eligibility.
What If You’re Not Sure Mesh Was Used?
Many patients do not remember whether mesh was implanted.
If you:
- Had complications
- Needed revision surgery
- Are currently discussing revision surgery
You may still qualify for review, even if you are unsure whether mesh was involved.
Medical records can help confirm whether mesh was used.
Why a Case Review Is Important
Every case is unique. Qualification depends on medical records, timing, complications, and surgical details.
If you believe you experienced serious complications after an internal bra or breast mesh procedure, speaking with an attorney can help you understand your options.
A professional review can determine whether your situation meets the criteria for a breast mesh lawsuit.
What Damages Could Be Recovered?
In legal terms, “damages” means losses caused by harm.
Possible damages in a surgical mesh lawsuit may include:
- Medical bills for revision surgeryHospital stays
- Antibiotics or infection treatment
- Lost wages from time off work
- Pain and suffering
- Emotional distress
- Future corrective procedures
Every case is different. That is why a personalized review matters.
What Should You Do If You Have Symptoms?
Prioritize health: contact your surgeon for pain, swelling, redness, or fluid buildup. Track records, seek second opinions, and report issues via FDA MedWatch. Discuss mesh use explicitly; early intervention often avoids explants or major revisions.
Think You May Have a Breast Mesh Claim?
If you suffered serious complications after an internal bra or surgical mesh breast procedure, you may have legal options.
The team at Vargas Gonzalez Delombard LLP is reviewing cases involving:
- Breast mesh infection
- Mesh migration
- Explant surgery
- Reconstructive failure
- Implant rupture
- Ongoing nerve pain
Do not wait. Time limits apply to filing claims.
Visit www.vargasgonzalez.com today to request a free consultation.
Your consultation is confidential. There is no obligation. And you pay nothing unless the firm recovers compensation for you.
If you believe mesh failure changed your life, now is the time to ask questions and protect your rights.





